 Sommerfeld postulated elliptical orbits that provided greater stability to the atoms, leading to the Bohr–Sommerfeld model. Due to the difficulties faced by Bohr’s model, A. The similarities of the textbooks published in four countries with different cultures and languages suggest that these textbooks have an underlying common thread, namely, the dominant empiricist epistemology. Exactly the same HPS-based criteria were also used to evaluate textbooks published in Turkey, Venezuela, and Korea (general physics). Bohr in general chemistry textbooks (published in the USA) revealed that most textbooks lack a historical perspective (although historical models are being presented) and provide a simplistic view of scientific models and how these change with no reference to the difficulties and controversies involved. Another study designed to evaluate the presentation of the atomic models of J. 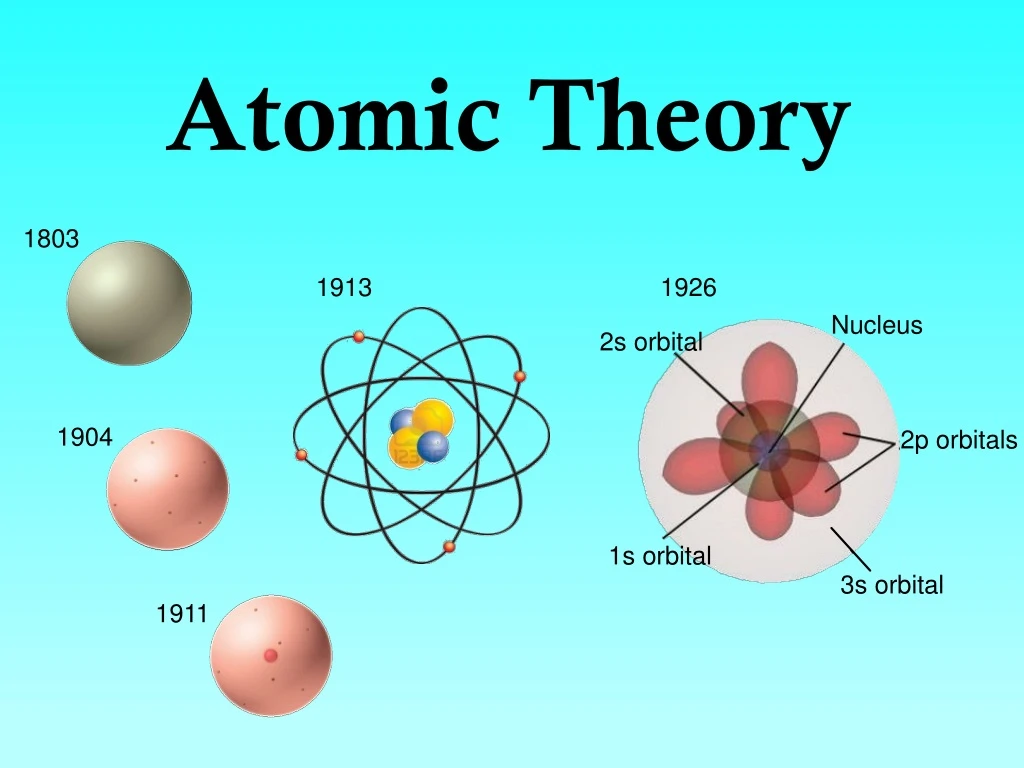
A study designed to evaluate the presentation of Dalton’s atomic theory in general chemistry textbooks (published in the USA) revealed that most textbooks stated that the atomic vision of Democritus was based on hypothetical questions (thought experiments), whereas Dalton based his theory on reproducible experimental results. On the contrary, Rocke (Email to author dated October 30, 2013, reproduced with permission 2013) considers that Dalton’s atomism is a successful theory. Dordrecht: Springer 2009) claims that Dalton’s theory had no testable content. Chalmers ( The scientist’s atom and the philosopher’s stone: How science succeeded and philosophy failed to gain knowledge of atoms. Dalton in developing the atomic theory is controversial among historians and philosophers of science. Understanding the role of early Greek philosophers (e.g., Democritus) and J.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
